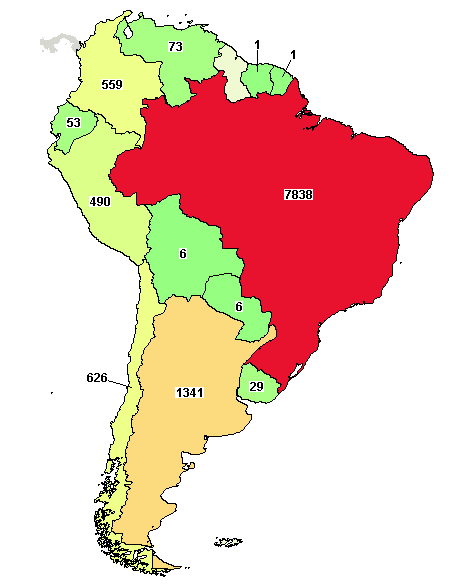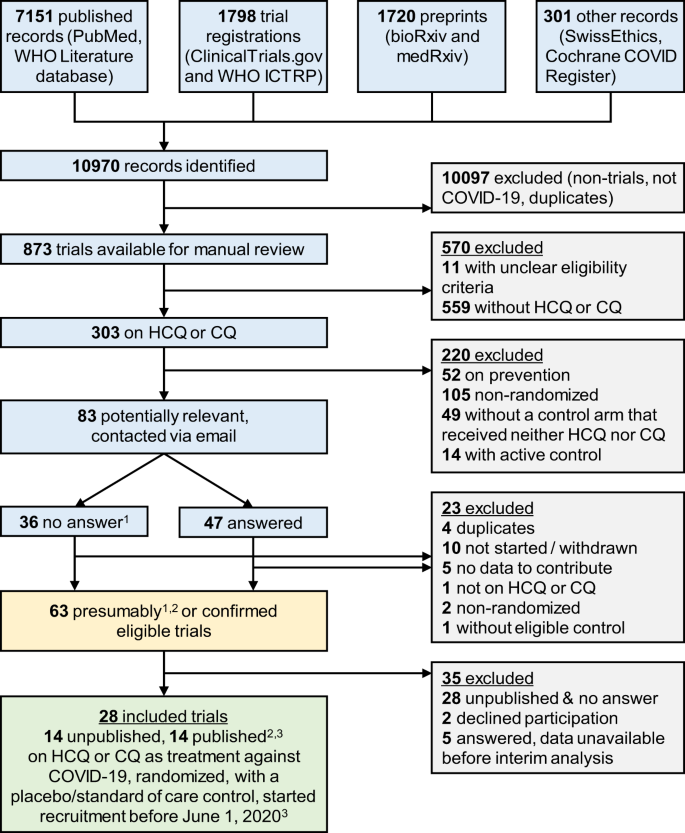
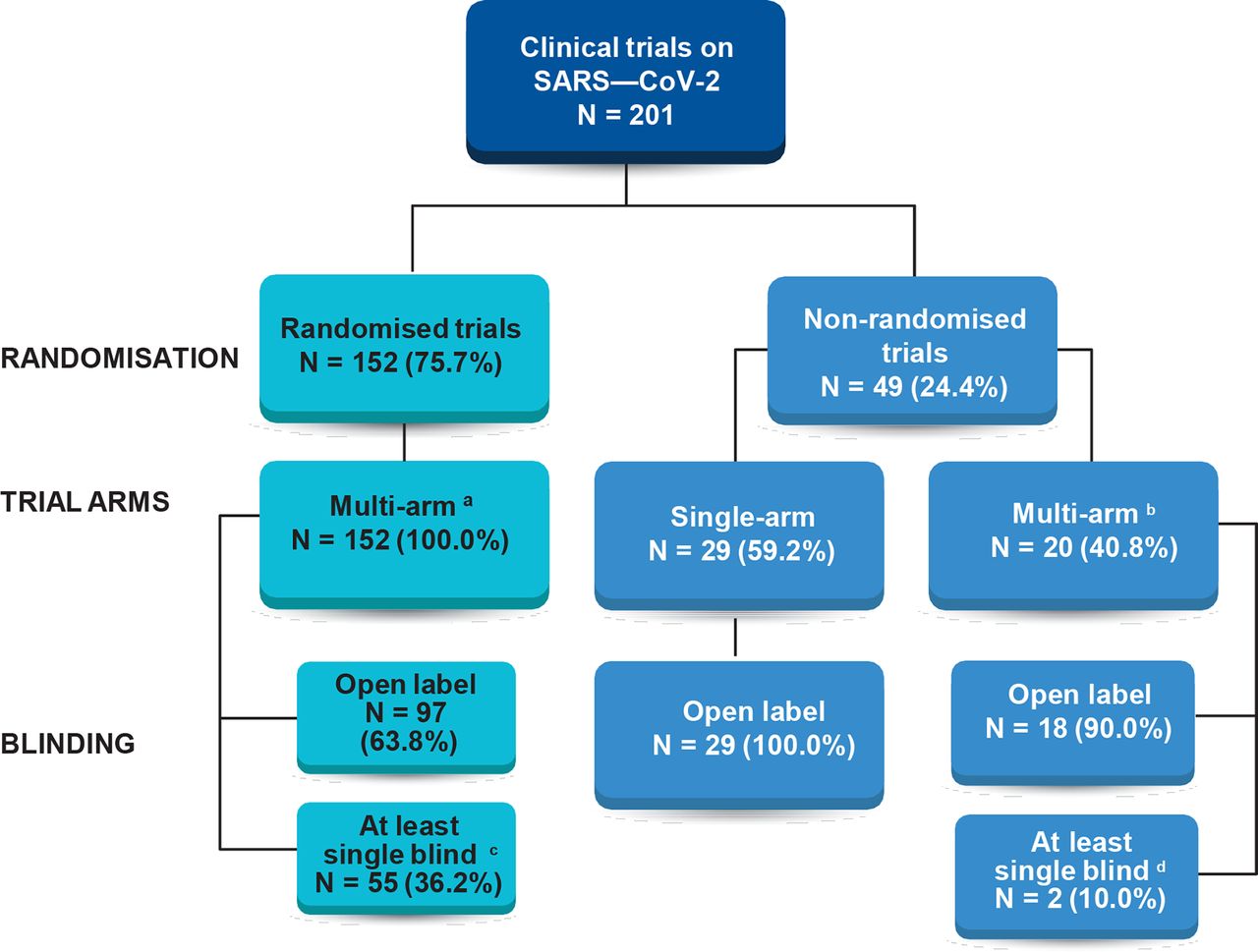
Mortality outcomes with hydroxychloroquine and chloroquine in COVID-19 from an international collaborative meta-analysis of randomized trials | Nature Communications
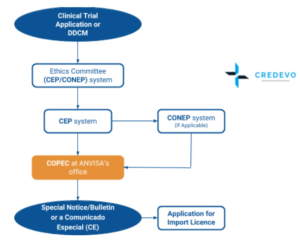
Characteristics of registered clinical trials assessing treatments for COVID-19: a cross-sectional analysis | BMJ Open

Trends in global clinical trial registration: an analysis of numbers of registered clinical trials in different parts of the world from 2004 to 2013 | BMJ Open

FACTORS RELATED TO THE COMPETITIVENESS OF THE COUNTRIES BRAZIL, ESTONIA AND MALAYSIA IN ATTRACTING GLOBAL CLINICAL TRIALS | Semantic Scholar

SciELO - Brasil - Avaliação de ensaios clínicos no Brasil: histórico e atualidades Avaliação de ensaios clínicos no Brasil: histórico e atualidades

Current updates on the European and WHO registered clinical trials of coronavirus disease 2019 (COVID-19) - ScienceDirect
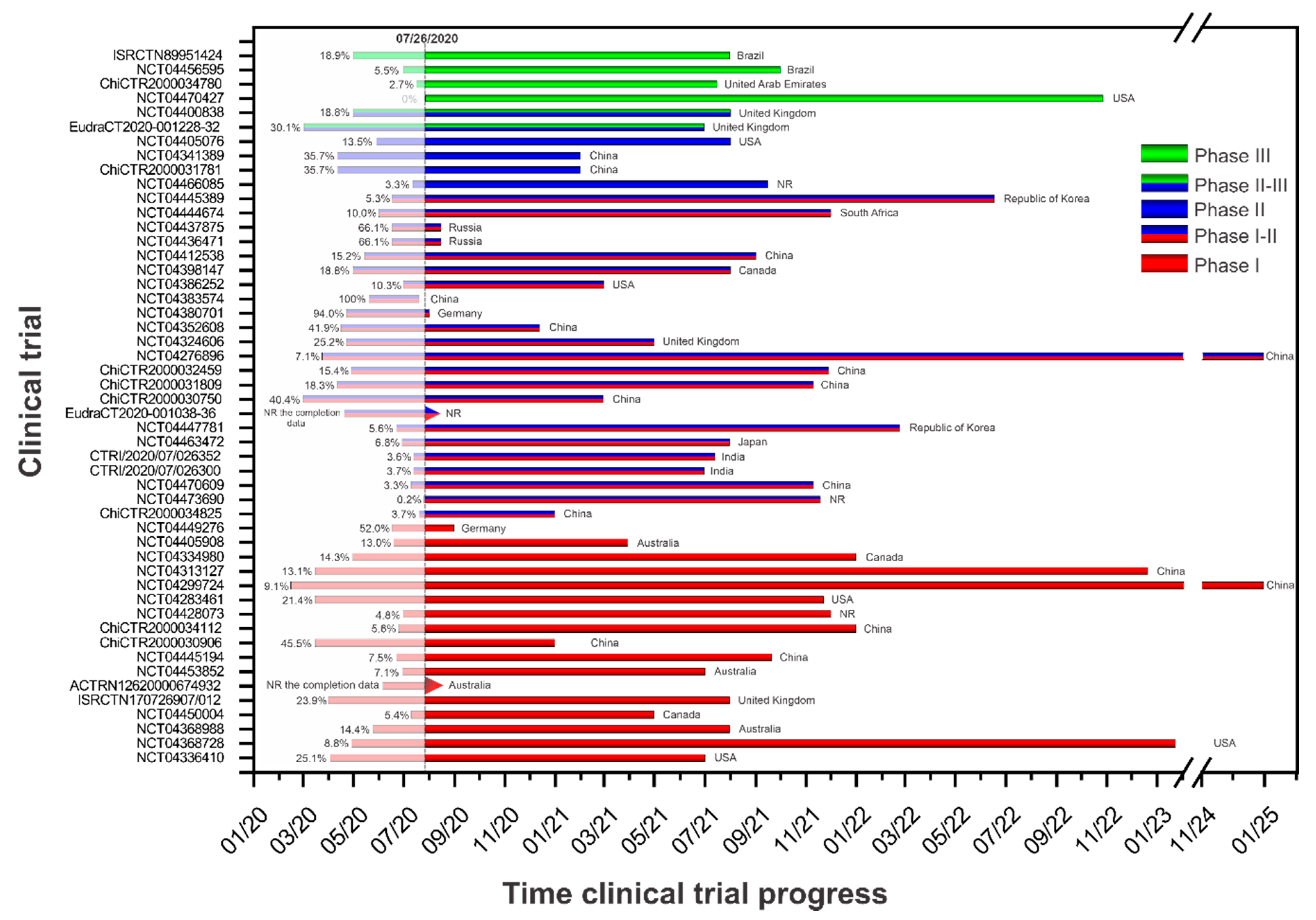
Vaccines | Free Full-Text | Current Clinical Trials Protocols and the Global Effort for Immunization against SARS-CoV-2 | HTML
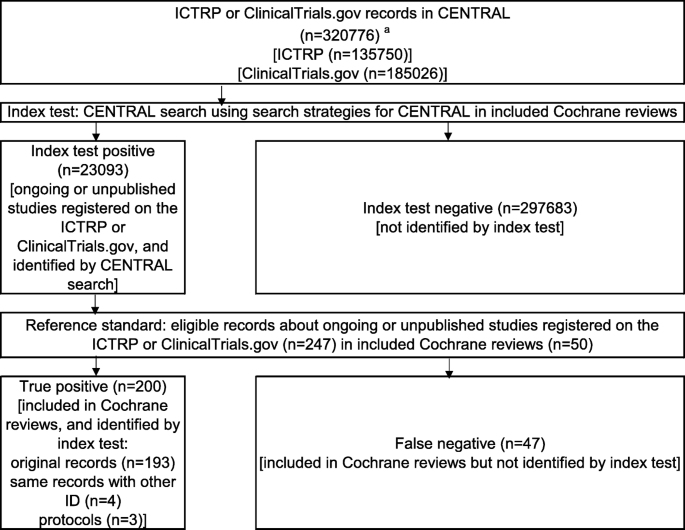
Using the Cochrane Central Register of Controlled Trials to identify clinical trial registration is insufficient: a cross-sectional study | BMC Medical Research Methodology | Full Text
GitHub - biomedical-translation-corpora/rebec: Clinical trials from the Brazilian Clinical Trials Registry - ReBEC

SciELO - Brasil - Clinical trials sponsored by industry and other private organizations Clinical trials sponsored by industry and other private organizations
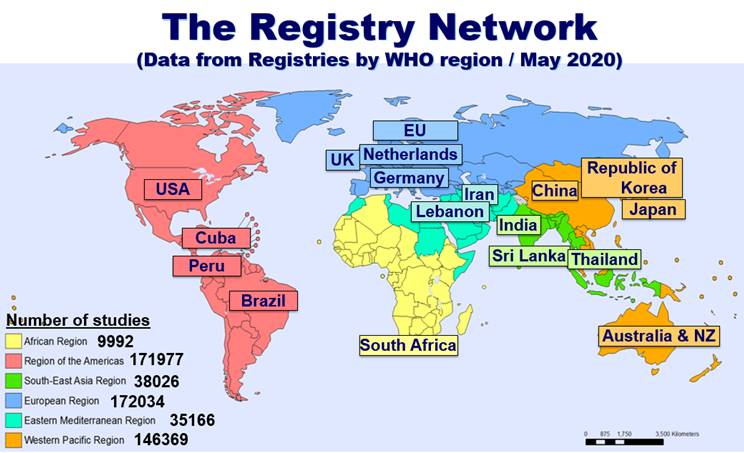
The WHO International Clinical Trials Registry Platform: Providing global clinical trial information to all - On Medicine

Quality assessment of clinical trial registration with traditional Chinese medicine in WHO registries | BMJ Open

Reporting of methods was better in the Clinical Trials Registry-India than in Indian journal publications - Journal of Clinical Epidemiology

Late-onset hereditary ATTR V30M amyloidosis with polyneuropathy: Characterization of Brazilian subjects from the THAOS registry - Journal of the Neurological Sciences

Cumulative number of COVID-19 clinical trials in Brazil according to... | Download Scientific Diagram

REGISTRY SNAPSHOT: Brazilian Clinical Trials Registry / Registro Brasileiro de Ensaios Clínicos (ReBEC) - Trial Assure

SciELO - Saúde Pública - Avaliação da resposta inicial de desenvolvimento de ensaios clínicos para COVID-19 no Brasil Avaliação da resposta inicial de desenvolvimento de ensaios clínicos para COVID-19 no Brasil