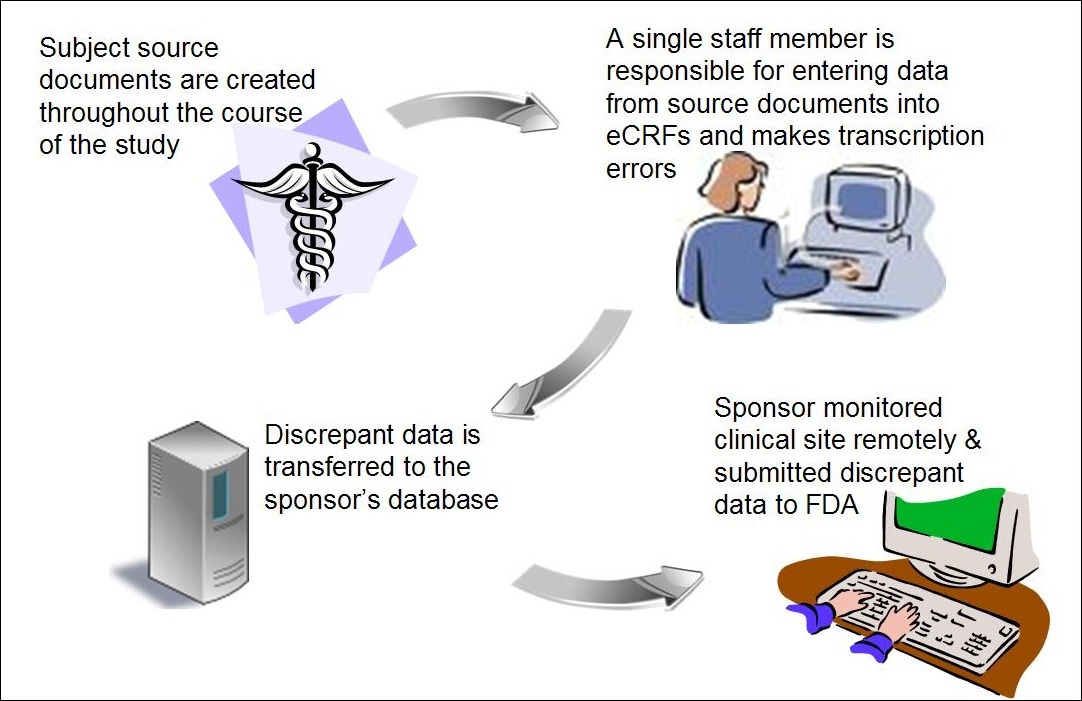
Medical Devices Clinical trials | How Electronic Data Capture (EDC) software contribute – ResearchManager
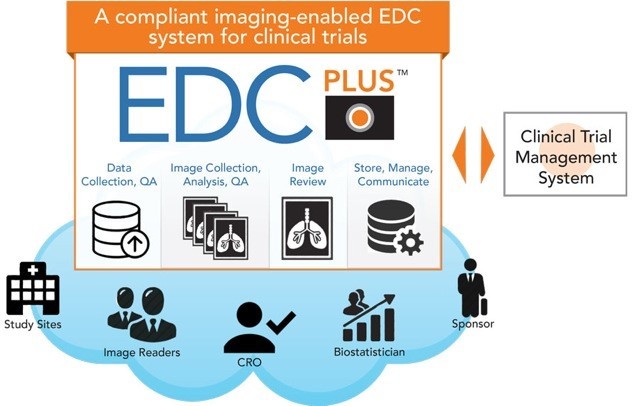
ImageIQ Releases EDCplus Technology, an Imaging-enabled Electronic Data Capture System for Clinical Trials | Business Wire

Electronic Source (eSource) vs. Electronic Data Capture (EDC): What's the Difference? - Clinical Research IO - CRIO